> Learn more about Cold Form vs. Thermoform Blister Packaging to make the right choice for your product.
Blister packaging does more than hold a pharmaceutical product like a tablet or capsule in place. It influences product protection, shelf life, and how reliably medication reaches the patient. At Pharmaworks, we work with both thermoform and cold form materials across many applications, and each option has its strengths depending on what the product requires.
The decision between thermoform and cold form blister packaging hinges entirely on the active ingredients’ sensitivity to environmental factors like light, moisture, and oxygen. Thermoform blister packaging utilizes heat to soften plastic films which are then vacuum-formed into custom pockets. Because these plastics are transparent, they allow for immediate visual inspection of the medication by pharmacists and patients. Thermoforming is ideal for stable chemical formulations, offers excellent line speeds, and minimizes material costs.
However, highly sensitive formulations, such as biopharmaceuticals, unstable antibiotics, or lyophilized tablets, degrade quickly when exposed to minimal ambient moisture or light. For these products, cold form blister packaging is required. This method mechanically presses a multi-layered aluminum laminate film into shape without heat. The resulting foil structure provides an absolute, near-impenetrable barrier against moisture, oxygen, and ultraviolet light, significantly extending product shelf life in challenging climates. While cold form packs require larger footprints and higher initial material investments due to the elasticity limits of aluminum, they serve as an essential insurance policy for delicate, high-value life science products.
🌡️ Thermoform Blister Packaging
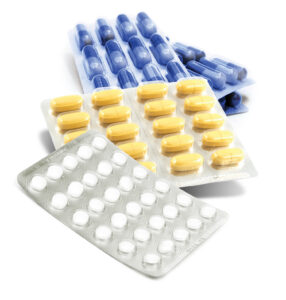 Thermoforming uses plastics such as PVC, PVDC, PCTFE, or PET. The film is heated until it becomes pliable, then shaped into cavities that match the product using air or plug assisted forming methods. After the product is placed into these formed pockets, the package is closed by sealing a lidding material to the top—typically a foil‑backed structure—to create a secure unit‑dose enclosure.
Thermoforming uses plastics such as PVC, PVDC, PCTFE, or PET. The film is heated until it becomes pliable, then shaped into cavities that match the product using air or plug assisted forming methods. After the product is placed into these formed pockets, the package is closed by sealing a lidding material to the top—typically a foil‑backed structure—to create a secure unit‑dose enclosure.
Thermoform Advantages
- Clear Visibility
The product can be seen through the cavity, which helps with identification. - Lower Material Cost
Thermoform films are generally more economical than cold form foil. - Fast Production
Thermoform lines typically run at higher speeds, which is ideal for large‑volume packaging. - Flexible Cavity Design
The material supports a wide range of shapes and sizes unlike cold formed aluminum. - Large Format and Deep‑Draw Capabilities
With the right material, thermoforming is well suited for larger or uniquely shaped pharmaceutical products.
Thermoform Limitations
-
- Moderate Barrier Performance
Thermoform films do not block moisture, oxygen, or light as effectively as cold form. - Shelf Life
Sensitive products may not maintain stability as long in a thermoformed cavity compared to cold form.
- Moderate Barrier Performance
❄️ Cold Form Blister Packaging
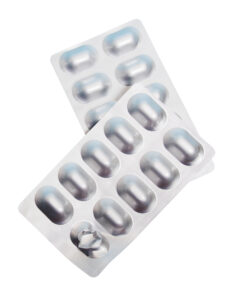
Cold form uses aluminum‑based material, commonly called “alu‑alu,” that is shaped mechanically using form plugs without the need for heat. This process produces a fully opaque, high‑barrier cavity suitable for products requiring maximum protection.
Cold Form Advantages
- Maximum Protection
Cold form provides a complete barrier against moisture, oxygen, and light. - Longer Shelf Life
Cold‑form foil delivers maximum barrier performance, helping sensitive products maintain stability and potency over extended shelf‑life requirements. - Regulatory Alignment
Often selected for critical medicines where long‑term stability is a priority.
Cold Form Limitations
-
- No Product Visibility
The cavity is opaque, so the product cannot be seen, resulting in reduced visual appeal and identification. - Higher Cost
Aluminum materials typically cost more than thermoform plastics, and the forming process is slower, leading to lower blister package output. - Limited Cavity Shapes
Because the material does not stretch, cavity designs are more restricted in both size and geometry. - Cavity and Blister Denting
Cold form blisters dent more easily than thermoformed materials due to the rigidity of aluminum. - Requires Additional Inspection
Achieving maximum barrier protection requires pinhole detection to ensure the aluminum does not develop micro‑tears during forming.
- No Product Visibility
⚖️ Key Comparison Table
| Feature | Thermoform Blister | Cold Form |
| Visibility | Transparent | Opaque |
| Barrier Protection | Moderate | Excellent |
| Shelf Life | Shorter | Longer |
| Cost | Lower | Higher |
| Production Speed | Faster | Slower |
| Design Flexibility | High | Limited |
How to Decide
If your product is not highly sensitive and you want clear presentation with efficient production, thermoforming is usually a straightforward choice. If the product needs maximum protection—especially from moisture or light—cold form is the safer option, even with higher material cost and slower output.
Pharmaworks equipment supports both formats, allowing you to match the packaging to the product rather than forcing the product into a single approach.
Blister Packaging Decision Checklist
- Product Sensitivity
- Highly sensitive to moisture, oxygen, or light → Cold Form
- Not highly sensitive → Thermoform
- Shelf-Life Requirements
- Extended shelf life (2+ years) → Cold Form
- Standard shelf life → Thermoform
- Visibility & Patient Appeal
- Need product visibility → Thermoform
- Visibility not required → Cold Form
- Cost Considerations
- Budget efficiency is a priority → Thermoform
- Higher material cost acceptable → Cold Form
- Production Speed
- Need high‑speed, large‑scale output → Thermoform
- Speed less critical → Cold Form
- Design Flexibility
- Require custom or complex cavity shapes → Thermoform
- Simple cavity geometry acceptable → Cold Form
- Regulatory & Safety Demands
- Product is critical, life‑saving, or highly regulated → Cold Form
- Standard regulatory profile → Thermoform
Quick Selection
- If your priorities lean toward protection, shelf life, and regulatory needs, cold form is likely the better fit.
- If your priorities lean toward visibility, cost efficiency, speed, and flexibility, thermoform is usually the better option.
For more Pharmaworks news, click here.
